
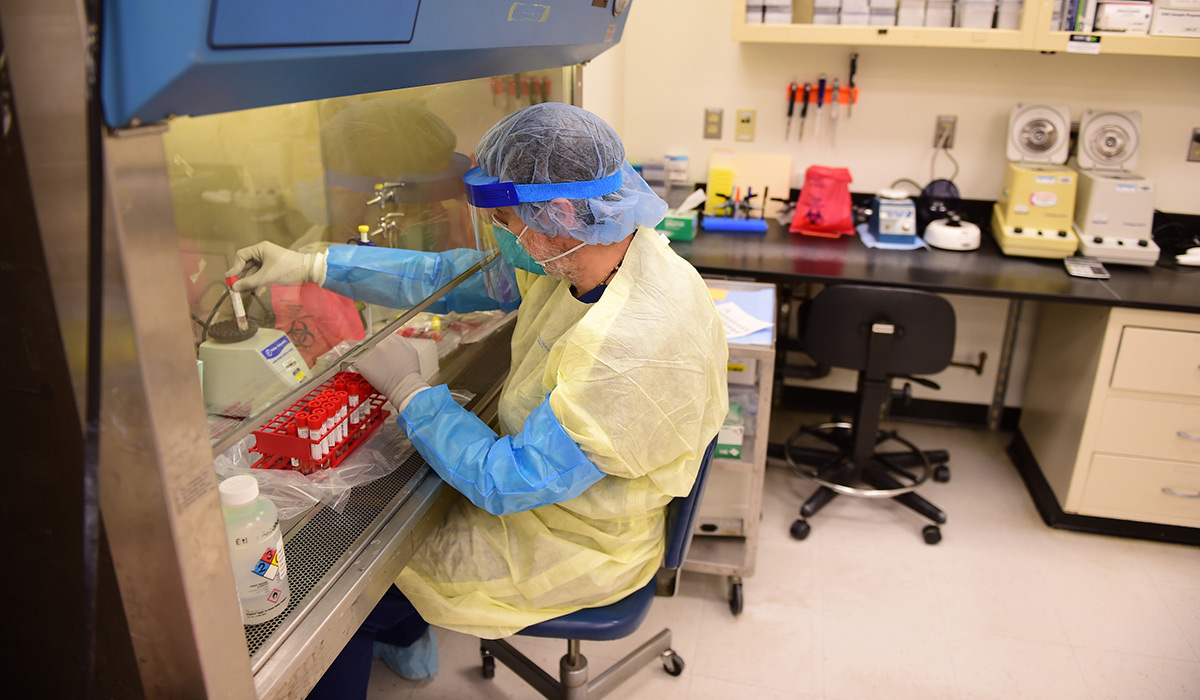
Joeffrey Chahine
While laboratory diagnostics are among the most important and frequently used tools for patient management and treatment at any time, work done by clinical laboratorians has become even more vital during the coronavirus pandemic. At MedStar health system — the largest healthcare system providing care in the greater Washington, D.C., and Baltimore region — students Joeffrey Chahine, Lauren Bassaro, and Masashi Waga, all pursuing doctoral degrees in Catholic University’s Clinical Laboratory Science Program, have played a major role in implementing laboratory assays to detect the virus that causes COVID-19 disease.
“Clinical laboratory scientists evaluate assays and make them operational for various clinical needs. In the case of the fight against COVID-19, time is a major factor,” said Wadad Al Salmi, clinical assistant professor and director of the program within the Department of Biology. “It was necessary to learn from global events starting early on so that actionable plans were evaluated and high-quality assays were acquired in the very tight supply chain market. These three students, who play different roles in the MedStar health system, greatly contributed in each step of evaluation and implementation.”

Lauren Bassaro, a medical technologist at MedStar Washington Hospital Center, is in her second year in the program. On weekdays, she goes to work at 5 a.m. to begin the first run of COVID tests before returning to Catholic University’s campus to conduct research on the protein rab17 and its role as a tumor suppressor in hepatocellular carcinoma.
In her Washington Hospital lab, a team of technologists has been working around the clock to run three varieties of tests for COVID-19. She says that the lab delivers approximately 200 tests for the virus each day.
“It definitely made my schedule crazy, especially while finishing up the semester,” Bassaro says. “I feel like I’ve been working myself thin.”
Bassaro became interested in clinical laboratory science as a way to conduct scientific research, while also helping others. Though her current work is rewarding, she also described it as a challenge because she is working with dangerous test samples.
“I feel like a lot of people forget about us because we’re in the basement of the hospital, but we have to work with these samples and we never know if they’re positive or negative until we get the results,” she said. “We’re still at risk, but we’re doing what we can, as fast as we can, to provide results.”
Joeffrey Chahine is a third-year doctoral student who works full time as the technical director for the molecular pathology division at MedStar Georgetown University Hospital. His laboratory team started working to begin diagnostic testing for COVID-19 in late-February. In early March, he was the first person in the MedStar system of 10 area hospitals to validate an open-ended molecular assay to be used for the detection of SARS-CoV-2 (COVID-19).
“Within the first two and a half weeks, we were able to test between 2,500 and 3,000 patients in my division,” Chahine said. “There were a lot of positive results unfortunately.”
Chahine and his team of eight technologists have been working seven days a week to continue the coronavirus tests, with each test taking about four and a half hours from process to result. This kind of rapid-result testing is quite rare in the world of molecular pathology, where results normally take two to three days to process.
“In my division, we can run up to 280 [SARS-CoV-2] tests per day,” Chahine said. In addition to the COVID-19 tests, his lab team also conducts molecular oncology and molecular hematopathology testing for various forms of cancers, as well as lymphoproliferative and myeloproliferative diseases such as leukemias and lymphomas.
The work is both physically and mentally draining. Because they work with the live virus, each technologist must wear specialized personal protective equipment (PPE), including full face and head covers, goggles, respirators, and face shields.
“Being there for four to five hours in these conditions is very exhausting, both mentally and physically, and especially with the PPE, after a while we feel like we can’t breathe,” Chahine said. “But this is what we signed up for, every single one of us. We’re glad we could step up at this time because if this process was implemented two to three weeks later, all these positive cases would have spread out even more throughout the area.”

Masashi Waga is MedStar Health’s technical director of microbiology operations. He has been working to assess and meet the demands for lab testing since late February. He also has been working closely with multiple vendors as well as healthcare leadership to obtain as many available assays as needed to ensure the MedStar system of hospitals could meet the high demand for COVID-19 testing.
“From the beginning, we knew that timing was very important,” Waga said. If his team didn’t act fast enough, there would be no assays available for purchase and area hospitals would have to rely on outside testing from large commercial laboratories.
“In the clinical operations, that would mean we would have to hold patients suspected to have COVID in the hospital for seven to 10 days,” he said. “If you don’t gain assays in-house, you have no choice but to occupy your entire hospital, making it impossible to take care of people who have other health conditions that need to be taken care of. That was really our goal — to prepare the worst-case scenario, so that we could avoid the total shutdown of our hospitals.”
Due to social distancing, which has resulted in a lower number of cases than were originally predicted, that healthcare system overload has not yet happened. Now the healthcare system is determining how hospitals can reopen for routine medical procedures while keeping both patients and staff safe.
“We expect patients to have some concerns coming back to hospitals,” he said. “Our strategy is to make more tests available in order to screen and manage patients as effectively as we can, which requires a different need for testing capacities.” In early May, he estimates that D.C. area hospitals are conducting between 600 and 800 tests per day, but that number is rising to include more asymptomatic patients.
“We are very proud of the work we’ve done. We were able to be resilient in this crisis because we were able to build test capability sufficiently,” Waga said. “I’m really appreciative of the teamwork we have between hospital entities and the different leadership teams, as well as the frontline lab teams who have really worked seamlessly to accomplish what is currently a pretty impressive testing capacity.”